ABSTRACT
The aerial parts of Panzeria alaschanica are used to treat pelvic inflammation and chronic pelvic inflammation. The anti-inflammatory effects of P. alaschanica have been well known in folk medicine, which suggest that extracted components from P. alaschanica could potentially treat inflammatory disease. The aim of this study was to discover the bioactive compounds of P. alaschanica. To achieve this, ethyl acetate (EtOAc) extract was extracted from the aerial parts of P. alaschanica and the anti-inflammatory effects were investigated against inflammation induced by carrageenan and egg-albumin in rats. At the doses used, EtOAc extract (100, 200 and 400 mg/kg) showed significant and dose-dependent anti-inflammatory effects. According to the high performance liquid chromatography (HPLC) analysis of EtOAc extract, there are two major families of compounds in the plant, namely phenylpropanoids (verbascoside,5,6,7,4'-tetrahydroxyflavone-7-O-(6"-O-[E]-coumaroyl)-β glucopyranoside,5,6,7,4'-tetrahydroxyflavone-7-O-(6"-O-[E]-caffeoyl)-β-glucopyranoside) and flavonoids (7,4'-dimethoxy-apigenin, isoramnetin-3-O-rutinoside, isoramnetin-3-O-glucoside, kaempferol-3-O-glucoside, hyperin), which may explain the activity.
Key words: Panzeria alaschanica, anti-inflammatory, high performance liquid chromatography (HPLC) analysis, phenylpropanoids, flavonoids.
Inflammation is a pathophysiological response of living tissues to injuries that leads to the local accumulation of plasmatic fluid and blood cells, which involves a complex sequence of bio-chemical events closely associated to the pathogenesis of various diseases, such as rheumatoid arthritis, osteoarthritis, ankylosing spondylitis, acute gout, and migraine (Vázquez et al., 2011; Kandati et al., 2012; Huang et al., 2011). Nowadays, although the synthetic anti-inflammatory drugs are dominating the market, the element of toxicity from these drugs cannot be ruled out. Due to the adverse reactions of synthetic and chemical medicines that is causing gastrointestinal irritation and reappearance of symptoms after discontinuation, herbal medicines have made a comeback to improve our basic health needs. Many plants and herbs, such as ginger, turmeric and olive oil, have been shown to exhibit potent anti-inflammatory effects (Gomase et al., 2011).
P. alaschanica, is a member of the Labiate family and distributed predominantly in Eerduosi of Inner Mongolia, China. P. alaschanica are one of the best-known traditional herbal medicines frequently used to treat pelvic inflammation and chronic pelvic inflammation (Li et al., 2011; Zhang et al., 2007). It is widely used in Mongolian as a substitute of the precious materia medica, Leonurus heterophyllus Sweet, which is used in treatment of edema, irregular menstruation, dysmenorrheal, amenorrhea and acute nephritis. However, there is few reported scientific study to support these claimed therapeutic and medicinal effects. During the recent years, flavonoids (Zhou and Bao, 2001; Hou et al., 2009) have been isolated from this plant, which were isolated from the ethyl acetate (EtOAc) extract of the aerial parts of P. alaschanica.
However, until recently, there have been few studies about the pharmacological effects of P. alaschanica. Recently, the anti-inflammatory effects of the EtOAc extract of P. alaschanica was studied by using egg- albumin-induced and carrageenan-induced rats. Moreover, a systematic chemical study was conducted on the EtOAc extract of P. alaschanica, which resulted in the isolation of phenylpropanoids and flavonoids (Hasan et al., 2012; Albacha et al., 2003; Meng et al., 2008; Julião et al., 2010) from the plant for the first time. This paper describes the HPLC analysis and the anti- inflammatory activity of the EtOAc extract of P. alaschanica.
Plant
The aerial parts of P. alaschanica, used as experimental material, were collected in Eerduosi, Inner Mongolia of China, in July 2013 and identified by Prof. Buhebateer (Inner Mongolia University for Nationalities). A voucher (NO. 20130722) was deposited in the School of Traditional Mongolian Medicine of Inner Mongolia University for Nationalities.
Extraction
The aerial parts of P. alaschanica (2.0 kg) was powdered and extracted by EtOAc (20 L) after extraction with 10 L chloroform (CHCl3). Evaporation of the solvent under reduced pressure resulted in the EtOAc extract (204.0 g). The dried extract was stored at 4°C before use.
Anti-inflammatory activity on carrageenan-induced paw oedema
The EtOAc extract was tested for anti-inflammatory activity on carrageenan-induced paw oedema, according to the method described by W inter et al. (1962). The animals were divided into eight groups of eight rats. The negative control group received distilled water (0.5 ml/kg, p.o.), the positive control group received luteolin (30 mg/kg, p.o.) and the test groups received the compounds at the doses of 100, 200 and 400 mg/kg p.o. The test was conducted using an electric plethysmometer 7140 (Ugo Basile, Italy). Carrageenan 2.5% (0.05 ml) was injected subcutaneously in the plantar surface of the rat’s left hind paw 1 h after oral administration of the drugs to induce a progressive swelling of the paw. The paw volume, up to the tibiotarsal articulation, was measured at 0 h (before carrageenan injection) and 1, 3, 5, 7, and 12 h later.
Egg-albumin-induced inflammation in rats
In testing for the effects of the compounds against inflammation, the method described by Albacha et al. (2003), was adopted. Briefly, rats were grouped into eight (n = 8). The negative control group received distilled water (10 ml/kg, p.o.), the positive control group received luteolin (30 mg/kg, p.o.) and the test groups received the EtOAc extract at the doses of 100, 200 and 400 mg/kg p.o. All the animals were injected with 0.1 ml of fresh egg- albumin subcutaneously into the left hind paw 30 min after the compounds and drug treatment. The volume of paw oedema of each rat was measured using a digital plethysmometer (LE 7500) prior to and 60 min after albumin injection and at every 60 to 300 min.
Acute toxicity
For the assessment of acute toxicity, W istar rats, male and female, were divided into groups of 10 animals. The EtOAc extract was given p.o. at the doses of 100, 500, 1000, 1500, 2000, and 3000 mg/kg from the first to seventh groups, respectively. The control group received p.o. distilled water (10 ml/kg). The mortality rate within 72 h period was determined and the LD50 was estimated according to the method described by references (Miller and Tainter, 1944).
Statistical analysis
Data were given as means ± standard error (SE); statistical analyses were done by using Student’s t-test. P < 0.05 is considered significant.
Sample preparation for HPLC analysis
About 1.0 g of the dried EtOAc extract was ground into powder, accurately weighed and soaked with 25 ml water and acetonitrile solution (70 : 30, v/v) for 60 min at room temperature, then sonicated for 20 min. The solution was filtered through a 0.45 µm membrane filter before LC analysis.
HPLC analysis and nuclear magnetic resonance (NMR) identification of HPLC fractions
The LC system consisted of an LC-20AT pump (Shimadzu, Japan), Shimadzu SPD-M20A photodiode array detector, Shimadzu CBM-20A software for data processing, and a Shimadzu CTO-22A column temperature box. The chromatographic separation was performed on Inertsil ODS-SP (250 mm × 4.6 mm, 5 µm). The binary gradient elution system consisted of water (A) and acetonitrile (B) and separation was achieved using the following gradient: 0 to 10 min, 15 to 25% B; 10 to 20 min, 25 to 50% B; 20 to 30 min, 50 to 65% B; 30 to 35 min, 65 to 75% B and finally, reconditioning steps of the column was 15% B isocratic for 5 min. The column temperature was kept constant at 30°C, 254 nm for monitoring, flow-rate of 1 ml/min and injection volume of 50 µl. The EtOAc solution was isolated by HPLC yielding AS1 (12 mg), AS2 (54 mg), AS3 (19 mg), AS4 (16 mg), AS5 (13 mg), AS6 (11 mg), AS7 (10 mg) and AS8 (10 mg).
The compounds obtained by HPLC were all identified by NMR. NMR spectra were measured on a Bruker AVAIVCE Ш-500 NMR spectrometer with tetramethylsilane (TMS) as the internal reference, and chemical shifts are expressed in δ (ppm).
Acute toxicity
No mortality was observed in groups of rats treated with the EtOAc extract of P. alaschanica. LD50 values for the EtOAc extract is more than 3000 mg/kg.
Anti-inflammatory activity
The development of oedema induced by carrageenan corresponded to the events in the acute phase of inflammation mediated by histamine, bradykinin and prostaglandins produced under an effect of cyclooxygenase. The EtOAc extract had an anti- inflammatory effect at 100, 200 and 400 mg/kg observable to 12 (p < 0.01), 7 (p < 0.01) and 4 h (p < 0.01), respectively (Figure 1A). The highest dose of EtOAc extract produced significant anti-inflammatory activity and the results were more than that of luteolin, while the activity of the medium dose is similar to luteolin. As is shown in Figure 1B, the EtOAc extract caused a significant and dose dependent inhibition of increase in paw edema. The highest dose of total flavonoids had higher anti-inflammatory effects than that of luteolin, while the activity of the lower dose is less than that of luteolin.
HPLC analysis and NMR identification of HPLC peaks
This developed HPLC assay method was applied to the analysis of the chemical constituents in the EtOAc extract obtained from P. alaschanica. The HPLC chromatogram is as shown in Figure 2, which mainly contained eight peaks and named as peaks 1 to 8. Sub- sequently, the peaks 1 to 8 were directly identified by the spectroscopic methods, including 1H NMR (Table 1), 13C NMR (Table 2) techniques, and by comparison with those reported in the literature (Hasan et al., 2012; Albacha et al., 2003; Meng et al., 2008; Julião et al., 2010).
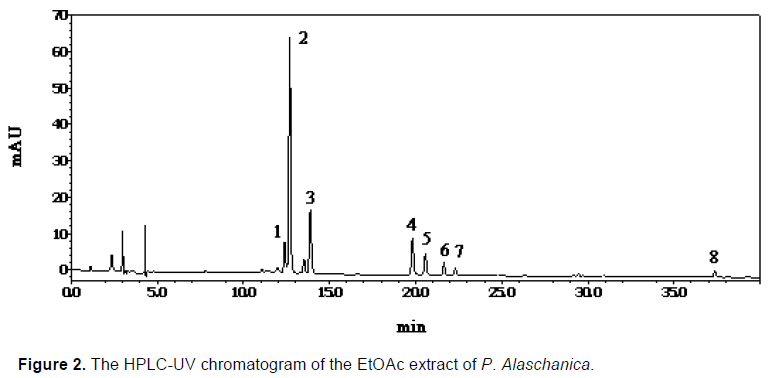
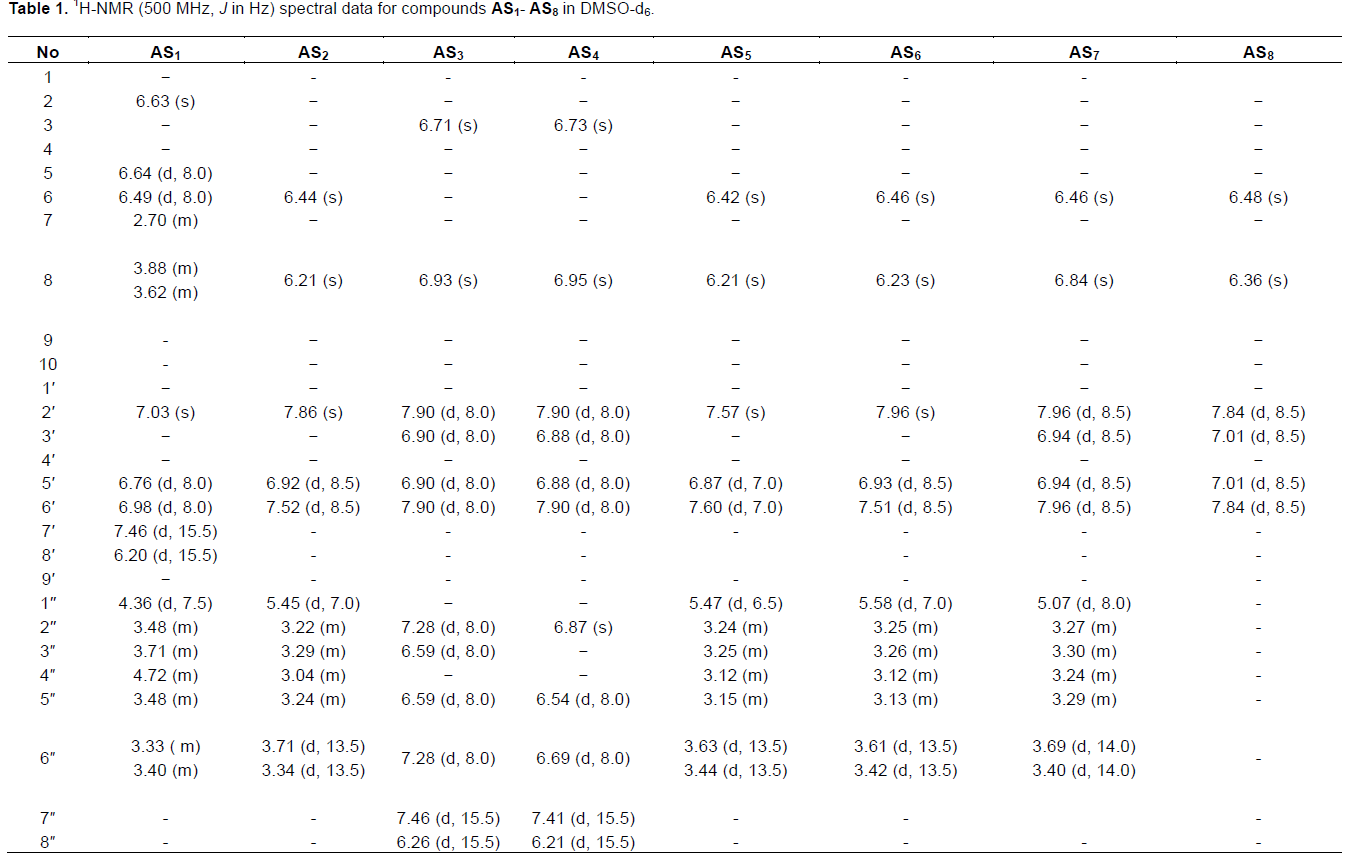
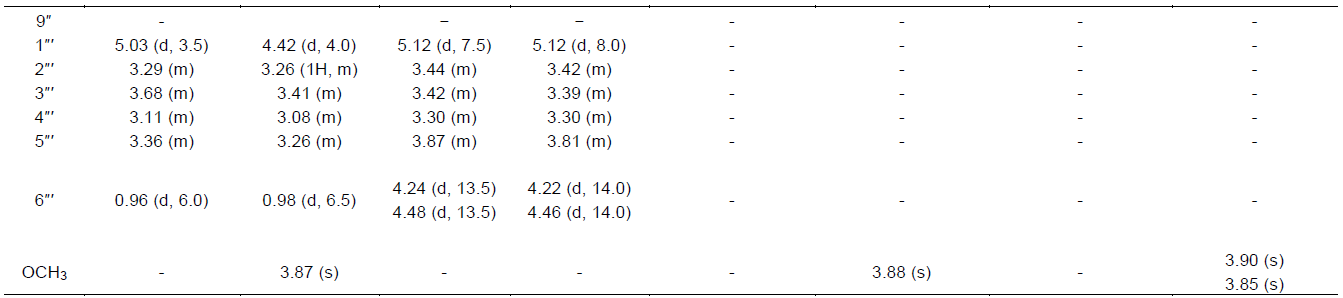
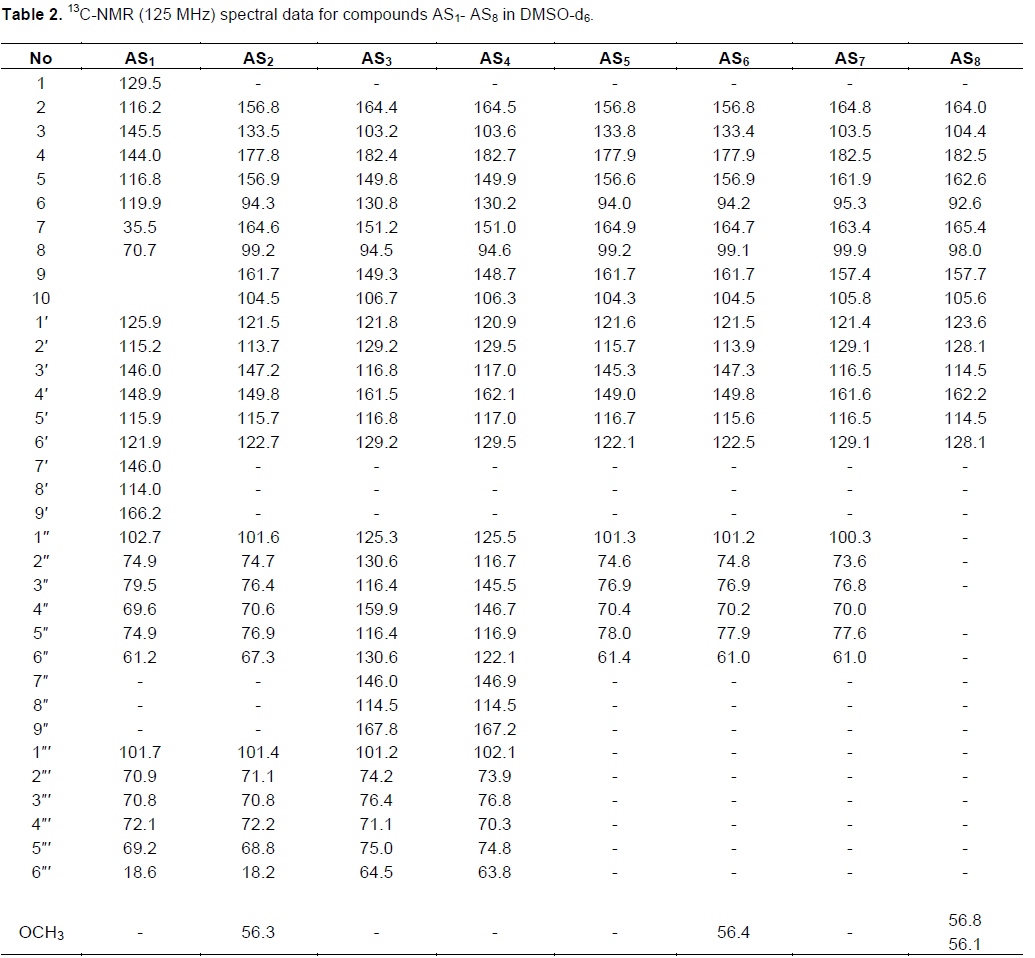
As a results, eight compounds (Figure 3) were isolated from the EtOAc extract, namely peak 1 (verbascoside, AS1), peak 2 (5,6,7,4'-tetrahydroxyflavone-7-O-(6"-O-[E]-coumaroyl)-β- glucopyranoside, AS2), peak 3 (5,6,7,4'- tetrahydroxyflavone-7-O-(6"-O-[E]-caffeoyl)-β- glucopyranoside, AS3), peak 4 (isoramnetin-3-O-rutinoside, AS4), peak 5 (isoramnetin-3-O-glucoside, AS5), peak 6 (hyperin, AS6), peak 7 (apigenin-7-O- glucoside, AS7) and peak 8 (7,4'-dimethoxy-apigenin, AS8).
The phenylpropanoids and flavonoids exhibited various biological properties like anti-bacterial, anti- inflammatory, and antioxidant and hepatoprotective effect (Peng et al., 2003; Ghisalberti, 2000; Silva et al., 2005), which may explain the anti-inflammatory activity of the EtOAc extract.
The present experimental investigation revealed that the EtOAc extract from P. alaschanica possessed significant anti-inflammatory activity in experimental animals at a dose of 400 mg/kg. Carrageenan-induced hind paw edema is the standard experimental model of acute inflammation. Moreover, the experimental model exhibits a high degree of reproducibility (Cadirci et al., 2012). It has a biphasic effect. The first phase is due to the release of histamine and serotonin (5-HT) (0-2 h), while plateau phase is maintained by a kinin like substance (3 h) and a second accelerating phase of swelling is attributed to prostaglandins (PG) release (>4 h) (Winter et al., 1962).
The EtOAc extract from P. alaschanica produced significant inhibition of carrageenan- induced paw edema. The inhibition was similar to that of the standard drug-luteolin. There are several mediators or multi processes underlining the pathogenesis of fever. Inhibition of any of these mediators may bring about antipyresis (El-Shenawy et al., 2002). On preliminary phytochemical screening, EtOAc extract from P. alaschanica was found to contain flavonoid compounds. Flavonoids are known to target prostaglandins, which are involved in the late phase of acute inflammation and pain perception (Morimoto et al., 1988). Our phytochemical study of the EtOAc extract from P. alaschanica revealed the presence of these compounds (verbascoside, 5,6,7,4'-tetrahydroxyflavone-7-O-(6"-O- [E]-coumaroyl)-β-glucopyranoside, 5,6,7,4' tetrahydroxyflavone-7-O-(6"-O-[E]-caffeoyl)-β- glucopyranoside, 7,4'-dimethoxy-apigenin, isoramnetin- 3-O-rutinoside, isoramnetin-3-O-glucoside, kaempferol- 3-O-glucoside, hyperin), which could be involved for antiinflammatory activity of the EtOAc extract. It has been also demonstrated that various flavonoids (7,4'- dimethoxy-apigenin, isoramnetin-3-O-rutinoside, isoramnetin-3-O-glucoside, kaempferol-3-O-glucoside, hyperin), phenylpropanoids (verbascoside, 5,6,7,4'- tetrahydroxyflavone-7-O-(6"-O-[E]-coumaroyl)-β- glucopyranoside, 5,6,7,4'-tetrahydroxyflavone-7-O-(6"- O-[E]-caffeoyl)-β-glucopyranoside) produced significant antinociceptive and/or anti-inflammatory activities (Hasan et al., 2012; Albacha et al., 2003; Meng et al., 2008; Julião et al., 2010). Hence, the presence of flavonoids and phenylpropanoids may be contributory to the anti-inflammatory activity of EtOAc extract from P. alaschanica. Although the exact nature of the anti- inflammatory activity mechanisms of the phytoconstituents have not been elucidated, the results of the present study were validated from a preclinical point-of-view, and the popular use of this medicinal plant in the treatment of inflammatory diseases. These studies are valuable for identifying lead compounds for anti-inflammatory drugs, keeping in mind the side- effects of NSAIDs and corticosteroids. Furthermore, human studies are needed to prove the safety and efficacy of long term administration of EtOAc extract from P. alaschanica as potential anti-inflammatory agent in routine clinical practice.
The authors have not declared any conflict of interests.
The authors thank the scientific research project of the Inner Mongolia Autonomous Region Universities in China (NJZZ14182) and the project of collaborative innovation center of Jiangxi Traditional Medicine (JXXT201402003). The authors are grateful to Ning Xu and Narenchaoketu for the measurements of NMR spectra.
REFERENCES
|
Albacha DC, Grayerb RJ, Jensen SR (2003). Acylated flavone glycosides from Veronica. Phytochemistry 64:1295-1301.
Crossref
|
|
|
|
Cadirci E, Süleyman H, Gürbüz P, Kuruüzüm UZA, Güvenalp Z, Demirezer LÖ (2012). Anti-inflammatory effects of different extracts from three Salvia species. Turk. J. Biol. 36:59-64.
|
|
|
|
|
El-Shenawy SM, Abdel-Salam OM, Baiuomy AR, El-Batran S, Arbid MS (2002). Studies on the anti-inflammatory and antinociceptive effects of melatonin in the rat. Pharmacol. Res. 46:235-243.
Crossref
|
|
|
|
|
Ghisalberti EL (2000). Lantana camara L. (Verbenaceae). Fitoterapia 71:467-486.
Crossref
|
|
|
|
|
Gomase PV, Shire PS, Nazim S, Choudhari AB (2011). Development and evaluation of polyherbal formulation for anti-inflammatory activity. J. Nat. Prod. Plant Resour. 1:85-90.
|
|
|
|
|
Hasan K, Etil A, Milena M (2012). Iridoid, phenylethanoid and flavonoid glycosides from Sideritis trojana. Fitoterapia 83:130-136.
Crossref
|
|
|
|
|
Hou FF, Zheng YF, Zhang HM, Shen SM, Du SS (2009). Chemical Constituents in Panzeria alaschanica Kupr. Chin. J. Exp. Tradit. Med. Form. 15:18-19.
|
|
|
|
|
Huang N, Rizshsky L, Hauck C, Nikolau BJ, Murphy PA, Birt DF (2011). Identification of anti-inflammatory constituents in Hypericum perforatum and Hypericum gentianoides extracts using RAW 264.7 mouse macrophages. Phytochemistry 72:2015-2023.
Crossref
|
|
|
|
|
Julião LDS, Leitão SG, Lotti C (2010). Flavones and phenylpropanoids from a sedative extract of Lantana trifolia L. Phytochemistry 71:294-300.
Crossref
|
|
|
|
|
Kandati V, Govardhan P, Reddy CS, Ravinder Nath A, Reddy RR (2012). In-vitro and in-vivo anti-inflammatory activity of Syzygium alternifolium (wt) Walp. J. Med. Plants Res. 6:4995-5001.
Crossref
|
|
|
|
|
Li XL, Zhang MF, Zhu D (2011). Extraction and determination of alkaloid in Mongolian herba Panzeria alaschanic. Chin J. Tradit. Chin. Med. Pharm. 26:2162-2164.
|
|
|
|
|
Meng YH, Krzysiak AJ, Durako MJ (2008). Flavones and flavone glycosides from Halophila johnsonii. Phytochemistry 69:2603-2608.
Crossref
|
|
|
|
|
Miller LC, Tainter ML (1944). Estimation of the LD50 and error by means of logarithmic probit graph paper. Proc. Soc. Exp. Biol. Med. 67:261-263.
Crossref
|
|
|
|
|
Morimoto A, Nakamori T, Watanabe T, Ono T, Murakami N (1988). Pattern differences in experimental fevers induced by endotoxin, endogenous pyrogen, and prostaglandins. Am. J. Physiol. 254:633-640.
|
|
|
|
|
Peng ZF, Strack D, Baumert A (2003). Antioxidant flavonoids from leaves of Polygonum hydropiper L. Phytochemistry 62:219-228.
Crossref
|
|
|
|
|
Silva GN, Martins FR, Matheus ME (2005). Investigation of anti-inflammatory and antinociceptive activities of Lantana trifolia. J. Ethnopharmacol. 100:254-259.
Crossref
|
|
|
|
|
Vázquez AI, Sánchez CM, Delgado NG, Alfonso AM, Ortega YS, Sánchez HC (2011). Anti-inflammatory and analgesic activities of red seaweed Dichotomaria obtusata. Braz. J. Pharm. Sci. 47:111-118.
|
|
|
|
|
Winter CA, Risley EA, Nuss GW (1962). Carrageenan-induced edema in hind paw of the rat as an assay for antiinflammatory drugs. Proc. Society Exp. Biol. Med. 11:544-548.
Crossref
|
|
|
|
|
Zhang YF, Yin W, Lin LL (2007). Advances in studies on Panzeria alaschanica. Chin. Tradit. Herbal Drug 38:1434-1436.
|
|
|
|
|
Zhou SX, Bao LR (2001). Studies on extracting and separating of flavonoids effective constituents in natural plant Panzerina. Acta. Univ. Inner. Mongol. 32:50-53.
|
|